
By the time the data is analyzed, and reports generated by the other CROs, it is too late to make any design modifications to a clinical trial. Our team can proactively plan and make any necessary changes much faster than CROs using traditional methods of data collection and reporting after database lock. Since we generate performance metrics, and assess subject enrollment/completion/screen failure rate/discontinuation throughout the life of a trial, we recognize study trends sooner. One of the reasons we recommend using Rave EDC is because the full Medidata platform functionality enables the study team to make decisions, perhaps even revise the protocol parameters and the clinical database, based on real-time subject data and trending. Several Rave EDC reports we develop are targeted to collate site and CRA performance metrics, such as whether there is a significant lag in site response or closure of queries issued by the system or manually generated by the study team. In addition to ad hoc reports, we use Rave EDC to schedule auto-generated reports for the convenience of our users. These ad hoc reports provide insight into the overall state of any of our clinical trials as well as site-specific statistics. With data extraction tools such as ODM Adapter and SAS on Demand, Rave EDC enables CDM to develop sophisticated reports using J-Review and Business Objects. 
With Rave EDC, we obtain clinical assessment data as well as subject/caretaker clinical reported outcomes (diaries), integrate these data into comprehensive reports about the clinical trial. This trend analysis capability of Rave CSA significantly improves Allucent’s efficiency and accuracy well in advance of database lock and statistical analysis.ĪLlucent CDM uses the industry leader in electronic data capture (EDC) technology, Medidata Rave EDC, to capture subject data from our investigative sites during a clinical trial. These same algorithms identify data clusters, look for data patterns, and generate rules for the data cluster any data not adhering to the rules are considered outliers and marked for evaluation. Rave CSA has machine learning algorithms to automatically detect and flag site entry errors for further review by CDM. 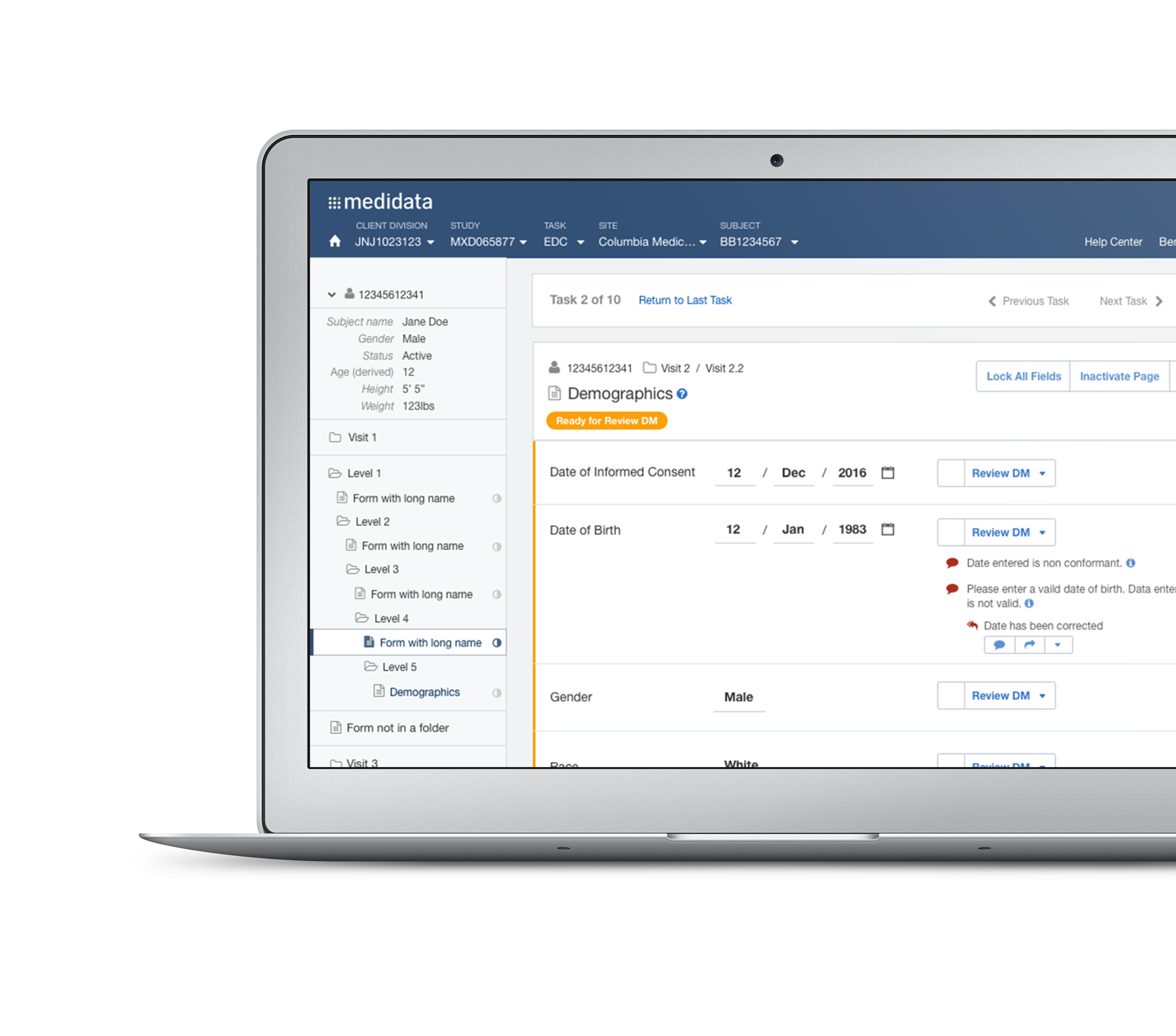
These real-time reports are easily accessible for all team members through a study portal showing site dashboards, tables, charts, or other visual graphics. With Medidata Rave CSA, Allucent CDM develops site-centric, safety focused reports to get on-demand snapshots of the subject lab values and other assessments to our Pharmacovigilance and Medical Monitoring Teams. Allucent uses this tool to transparently track site performance based on the timeliness and accuracy of a site’s data entry. Medidata’s Rave Centralized Statistical Analytics (Rave CSA) facilitates real-time review and global analysis of subject data across all regions of the world. In this post, we cover how Allucent leverages all of them to achieve a reliable and timely database lock, the Allucent way.ġ) RAVE CENTRALIZED STATISTICAL ANALYTICS Allucent has several tools at our disposal to clean and report on this data in an ongoing manner to help the full study team identify and mitigate any risk decisively: Having a solid CDM strategy ensures statistically sound data that enables stakeholders to develop sound, fact-based conclusions from subject data.Īt Allucent, our years of experience in conducting all kinds of clinical trials underlines the importance of knowing the CDM process starts from the instant the first piece of data is received from our sites in the Electronic Data Capture (EDC) platform. CDM ensures all clinical data is handled not only in accordance with current global regulatory standards but meets the highest standards of Good Clinical Practice (GCP ICH). A critical component to the overall success of any clinical trial is how Clinical Data Management (CDM) manages the trial during its conduct and how it closes it out to achieve database lock.Īs you know, CDM is the process of collecting, analyzing, and integrating vast volumes of clinical data together to paint a picture of an investigative product or device safety and efficacy for our clients. Your clinical trial has been running for years potentially and now it is time to achieve a database lock that is efficient, quality driven and on time. DMPK and Translational Discovery and Development.Pharmacometrics (PMx), Pharmacokinetics, Pharmacodynamics (PK/PD).Clinical Pharmacology Integrated Drug Development.Clinical Pharmacology Modeling Simulation.Biostatistics and Statistical Programming.Chemistry, Manufacturing, and Controls (CMC).Regulatory and Drug Development Consulting.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
